Lume Innovations
The Virridy Lume addresses many of the limitations identified in the TLF literature by coupling optical TLF sensing with machine learning models that account for environmental confounders. Rather than relying on static TLF thresholds, the Lume's algorithm adapts to site-specific conditions.
60s
Instant Results
The Lume returns a TLF reading in 60 seconds, compared to 24–48 hours for traditional culture-based methods.
< 0.1 ppb
Detection Limit
The Lume's minimum detection limit for tryptophan dissolved in deionized water.
1 CFU
Regulatory Threshold Classification
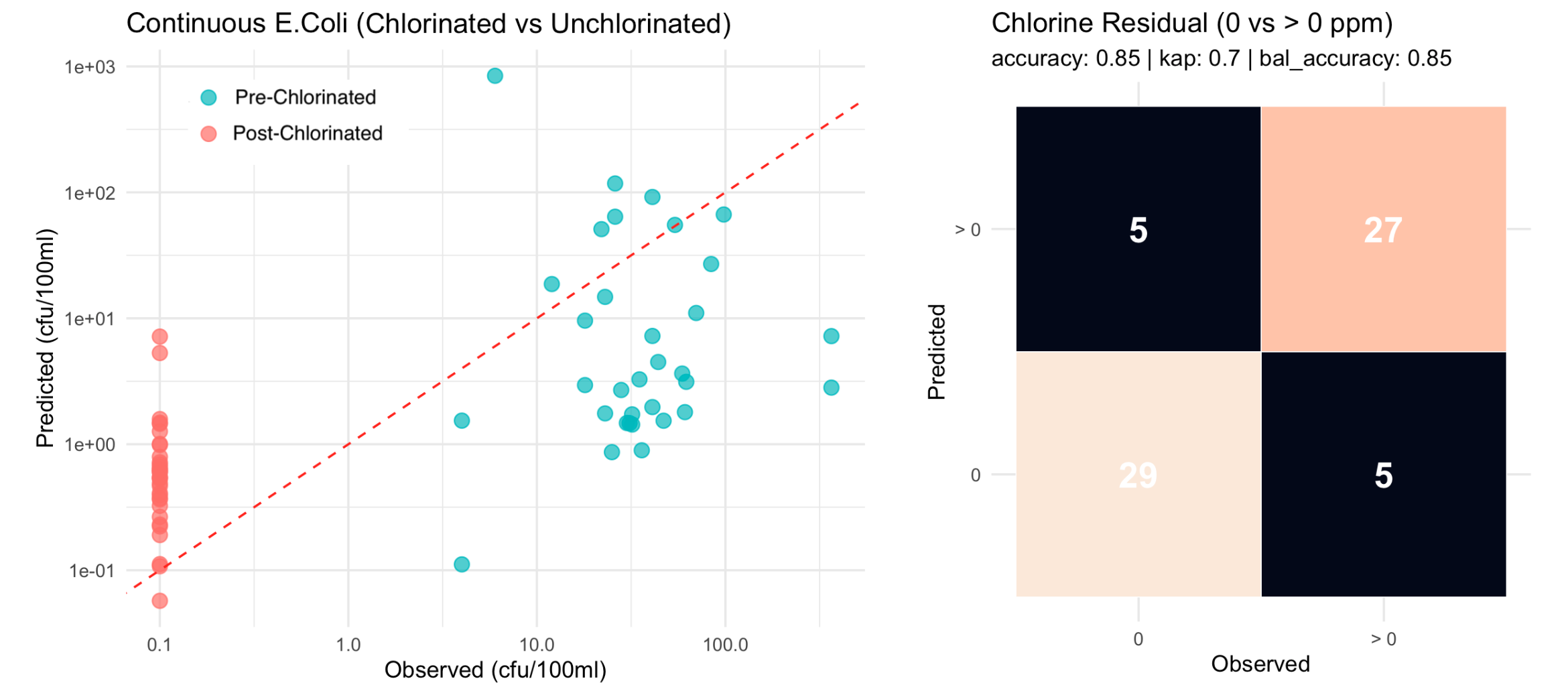
The Lume's binary classification at the 1 CFU/100 mL drinking water regulatory threshold yields 91% overall accuracy (Cohen's kappa = 0.82).
>94%
Categorical Accuracy
Site-specific calibrated categorical classifications of microbial contamination risk.
75%+
Linear Accuracy
Out-of-the-box accuracy on a continuous scale across 0–1,000 CFU/100 mL.
7%
Log-Scale Error
Mean absolute percentage error in log-transformed concentration space vs. culture-based methods.
Key Innovations
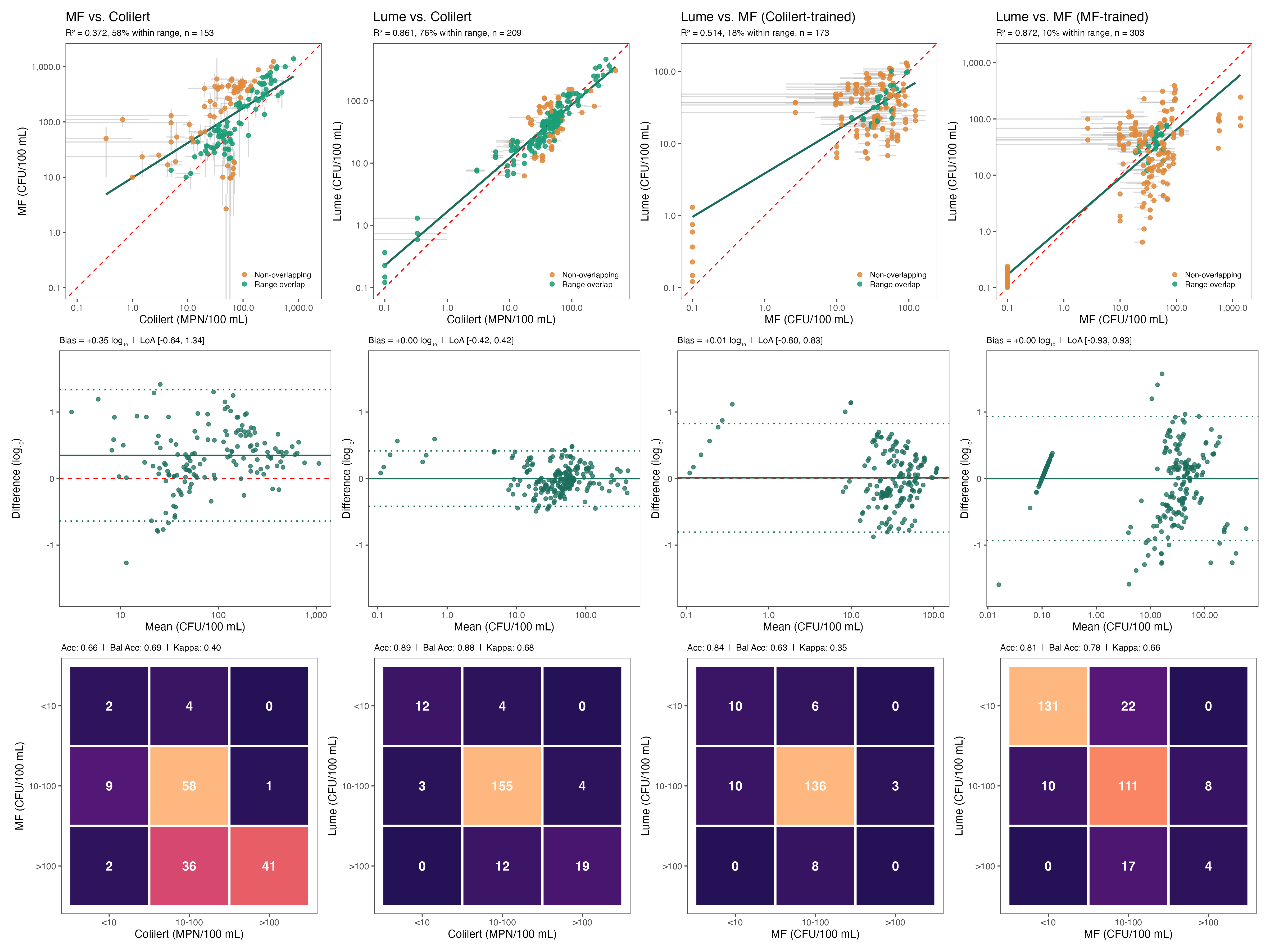
Machine learning quantification: Unlike earlier TLF sensors that report relative fluorescent units, the Lume quantifies actual E. coli concentrations using gradient-boosted decision tree models that capture nonlinear patterns in noisy sensor data. The ML model was validated across freshwater and coastal environments, including saline settings where enterococci are the regulatory indicator.
Multi-parameter correction: Built-in turbidity and temperature sensors allow the algorithm to correct for environmental interference that confounds raw TLF readings.
Drinking water performance: Binary classification at regulatory thresholds (1 and 10 CFU/100 mL) achieves 91–92% overall accuracy with Cohen's kappa of 0.82–0.84, indicating strong agreement with laboratory classifications.
Ocean transferability: In initial ocean deployments, the Lume achieved over 76% categorical accuracy using just six training samples, demonstrating transferability across microbial indicators and water matrices.
Patented Technology
Adaptive contamination detection: The Lume uses machine learning to learn normal conditions and trigger contamination alerts based on learned patterns rather than fixed thresholds. (US Patent 11,506,606 B2 — Bedell, Fankhauser, Sharpe, Wilson & Thomas)
Automated system-state classification: Time-series data from water infrastructure sensors are analyzed to classify system states and support operational decisions without manual inspection or rule-based logic. (US Patent 11,507,861 B2 — Wilson, Coyle, Thomas & Croshere)